Atai Life Sciences
Atai’s cash levels fall as company assures investors it has enough money
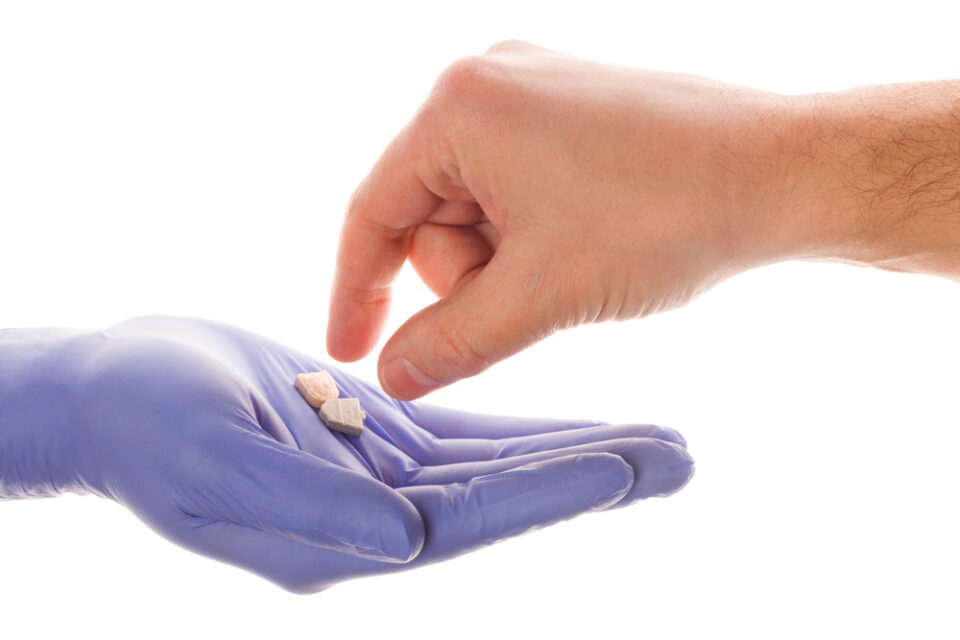
Atai Life Sciences
Atai extends cash runway into 2027 despite $39M fourth-quarter loss
Atai Life Sciences
Atai Life Sciences prices $55 million offering
Atai Life Sciences
Atai, Awakn make progress on treatments for alcohol use disorder
-

 California Cannabis Updates1 year ago
California Cannabis Updates1 year agoAlert: Department of Cannabis Control updates data dashboards with full data for 2023
-

 Breaking News1 year ago
Breaking News1 year agoConnecticut Appoints The US’s First Cannabis Ombudsperson – Yes there is a pun in there and I’m Sure Erin Kirk Is Going To Hear It More Than Once!
-

 best list1 year ago
best list1 year ago5 best CBD creams of 2024 by Leafly
-

 Business1 year ago
Business1 year agoRecreational cannabis on ballot for third time in South Dakota
-

 Business1 year ago
Business1 year agoEU initiative begins bid to open access to psychedelic therapies
-

 cbd1 year ago
cbd1 year agoNew Study Analyzes the Effects of THCV, CBD on Weight Loss
-

 Bay Smokes1 year ago
Bay Smokes1 year agoFree delta-9 gummies from Bay Smokes
-

 autoflower seeds1 year ago
autoflower seeds1 year ago5 best autoflower seed banks of 2024 by Leafly














































